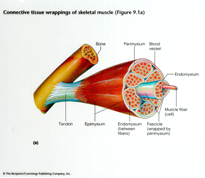
Skeletal Muscle
Store-operated calcium entry in muscle health, aging and fatigue
During aging skeletal muscles become weaker and hundreds of
millions of individuals become afflicted by a condition named sarcopenia. While some of the changes that take place in aged
skeletal muscles are directly related to muscle atrophy, changes
in contractile apparatus function cannot fully explain the
decrease in the specific force during aging. Our recent studies
revealed that muscle aging is associated with compromised
store-operated calcium entry (SOCE, see figure below), the
elemental process for refilling of the intracellular calcium
stores in skeletal muscles and the majority of cells studied to
date. As calcium (Ca) entry is chronically reduced in aged
muscle fibers, Ca storage in the muscles decrease making less Ca
available for each contraction-relaxation cycle, leading to
reduced contractile force and fatigue. Thus, my lab was among
the first to demonstrate that effective SOCE is essential for
normal muscle contractility. Our research involves the use of
genetically modified mouse models, molecular-genetic
manipulations, muscle contractility studies, calcium imaging and
a number of biochemical and biophysical approaches. We always
combine in vitro with in vivo approaches to
create a broader understanding of physiological phenomena. A
number of our studies are now being expanded to humans' patients
with different muscle diseases.
Excitation-contraction (E-C) coupling
E-C coupling, illustrated in the figure below,
is a cellular signaling process by which a depolarization signal
from a motor neuron results in the contraction of a skeletal
muscle fiber. Contraction is induced by a global release of
calcium from the sarcoplasmic reticulum (SR) into the cytoplasm
through the ryanodine receptor calcium release channel. Using an
extensive immuno-proteomic approach, our research group in
collaboration with Drs. Jianjie Ma at Robert Wood Johnson Medical
School and Hiroshi Takeshima at Kyoto University has identified
and characterized the function of several novel genes of the
molecular machinery that regulates E-C coupling in muscle under
physiologic and pathophysiologic conditions. In addition, in
collaboration with Drs C-K Qu and Thomas Nosek at Case Western
Reserve University and Dr. Hector Valdivia, University of
Wisconsin, we have discovered a novel muscle specific
phosphatase (MIP/MTMR14) that is essential for normal muscle
function. Recent studies have demonstrated that mutations in the
MIP gene relate to centronuclear myophathies and Charcot-Marie
Tooth Syndrome, two devastating muscle diseases. Our long-term
goal is to translate what we learn in the lab to the clinical
environment to improve the lives of animals and humans suffering
from muscle diseases.
Laboratory Members
Leticia Brotto, MD, Research Associate & Lab Manager
Sandra Romero-Suarez, PhD, Postdoctoral Research Associate
Todd Hall, BSA, Research Associate
Cheng Lin Mo, MS, Graduate Student Research Assistant
Michael Loghry, EMT, Undergraduate Research Assistant
Kendra Baker, BA, Research Assistant